Deprecated: Creation of dynamic property InsertPostAds::$settings is deprecated in /home4/keralaon/public_html/wp-content/plugins/insert-post-ads/insert-post-ads.php on line 427
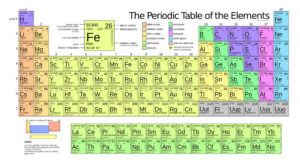
Periodic Table – periodic table in chemistry
The periodic table is a tabular arrangement of the chemical elements, ordered by their atomic number (number of protons), electron configurations, and recurring chemical properties. This ordering shows periodic trends , such as elements with similar behaviour in the same column. It also shows four rectangular blocks with some approximately similar chemical properties. In general, within one row (period) the elements are metals on the left, and non – metals on the right.
The rows of the table are called periods ; the columns are called groups. Six groups have names as well as numbers. The periodic table can be used to derive relationships between the properties of the elements, and predict the properties of new elements yet to be discovered or synthesised. The periodic table provides a useful framework for analysing chemical behaviour, and is widely used in chemistry and other sciences.
Periodic Table